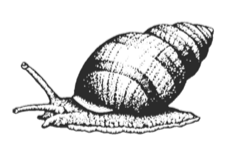
Littorina spp. are small intertidal snails (Phylum Mollusca, Class Gastropoda). There are five species of littorines in the Pacific Northwest, including: Littorina plena, L. scutulata, L. sitkana, L. subrotundata and L. littorea. L. littorea is an introduced species.
Description:
Littorines, like all gastropods, have 3 body regions: A well-developed head, a visceral mass, and a muscular foot. The head and foot extend out from a coiled shell when the animal is moving or exploring the environment. The visceral mass that consists of the body organs is tucked inside the blue-black shell that has a maximum length of 52 mm from tip to the opening (Jackson 2008). L. littorea is the largest littorine, as other littorines rarely have a shell height greater than 18 mm (Jackson 2008).
Littorines detect environmental conditions with sensory organs located on the head. Two simple eyes detect light intensity while two pair of tentacle detect odors or feel the environment (Carefoot n.d.). Littorines also use their tentacles to detect the mucus trails left by other littorines (Carefoot n.d.).
Beneath the shell a large muscular foot contracts to move the animal along the substratum (University of Rhode Island n.d.). The foot contains a specialized organ, called an otocyst, which the snail uses to maintain balance. Mucous produced by a gland at the anterior end of the foot coats the bottom of the foot and eases movement across rough surfaces (Carefoot n.d.). The foot can also form suction to hold the animal in place to resist the force of waves (Carefoot n.d.). If threatened or stressed by environmental conditions littorines can use a small piece of shell attached at the end of the foot, called the operculum ("little lid") to close the opening of the shell. If conditions are extreme they can also secrete a special mucus around the opening to their shell (University of Rhode Island n.d.). When the mucous hardens it cements the snails onto the substratum where they will wait until conditions improve (University of Rhode Island n.d.).
Littorines detect environmental conditions with sensory organs located on the head. Two simple eyes detect light intensity while two pair of tentacle detect odors or feel the environment (Carefoot n.d.). Littorines also use their tentacles to detect the mucus trails left by other littorines (Carefoot n.d.).
Beneath the shell a large muscular foot contracts to move the animal along the substratum (University of Rhode Island n.d.). The foot contains a specialized organ, called an otocyst, which the snail uses to maintain balance. Mucous produced by a gland at the anterior end of the foot coats the bottom of the foot and eases movement across rough surfaces (Carefoot n.d.). The foot can also form suction to hold the animal in place to resist the force of waves (Carefoot n.d.). If threatened or stressed by environmental conditions littorines can use a small piece of shell attached at the end of the foot, called the operculum ("little lid") to close the opening of the shell. If conditions are extreme they can also secrete a special mucus around the opening to their shell (University of Rhode Island n.d.). When the mucous hardens it cements the snails onto the substratum where they will wait until conditions improve (University of Rhode Island n.d.).
Ecology:
Littorines have a worldwide distribution, but are more common in northern temperate and subarctic regions (Carefoot n.d.). They primarily inhabit the mid and high intertidal regions of rocky intertidal zones, but they may also be found in salt marshes and mudflats (Encyclopedia Britannica Editors 2008; Jackson 2008). When the tide is low, they are commonly found under macroalgae and in rock crevices or they take refuge in empty barnacle shells.
As littorines move across the substratum they scrape food off the surface using a saw-toothed radula tucked in the mouth. They primarily consume diatoms and other microalgae and lichens (Carefoot n.d.). Littorines will stop feeding and either hide in cracks or draw in their operculum when threatened by predators such as crabs, sea stars, carnivorous gastropods, fishes, and birds (Carefoot n.d.).
As littorines move across the substratum they scrape food off the surface using a saw-toothed radula tucked in the mouth. They primarily consume diatoms and other microalgae and lichens (Carefoot n.d.). Littorines will stop feeding and either hide in cracks or draw in their operculum when threatened by predators such as crabs, sea stars, carnivorous gastropods, fishes, and birds (Carefoot n.d.).
Life History:
Littorines can live 5 to 10 years (Jackson 2008). In the spring mature males will fertilize the females. Then depending upon the species, either the egg cases are released directly into the water (L. scutulata, L. plena, and L. littorea) or they are attached to the substratum (L. sitkana and L. subrotundata). In those species with pelagic egg cases, the embryos develop into free-swimming larvae that drift in the water (Carefoot n.d.). They eventually settle to the substratum, but can delay settlement by the production of mucus threads that reduces the rate of sinking to the bottom (similar to how juvenile spiders use a silken thread to disperse by air) (Carefoot n.d.). In those species with benthic egg cases, the embryos develop directly into juvenile snails that disperse to new habitat by crawling to a new site (Carefoot n.d.).
Referenced cited:
Carefoot T. n.d. A Snail’s Odyssey. Learn about Littorines & Relatives. [accessed 2017 May 17]. http://www.asnailsodyssey.com/littorine.php .
Encyclopedia Britannica Editors. 2008. Periwinkle, Marine Snail. Encyclopedia Britannica, Inc. [accessed 2017 May 17]. https://www.britannica.com/animal/periwinkle-marine-snail.
Jackson A. 2008. Littorina littorea: Common periwinkle. In: Tyler-Walters H, Hiscock K, editors. n.d. MarLIN: Marine Life Information Network. [accessed 2017 May 17]. http://www.marlin.ac.uk/species/detail/1328.
University of Rhode Island: Environmental Data Center. n.d. Common Periwinkle : Littorina Littorea. [accessed 2017 May 17]. http://www.edc.uri.edu/restoration/html/gallery/invert/peri.htm.
To learn more:
Carlson RL, Shulman MJ, Ellis JC. 2006. Factors contributing to spatial heterogeneity in the abundance of the common periwinkle Littorina littorea. Journal of Molluscan Studies. 72(2): 149-156.
Davenport J, Davenport JL. 2005. Effects of shore height, wave exposure and geographical distance on thermal niche width of intertidal fauna. Marine Ecology Progress Series. 292: 41-50.
Davies MS, Beckwith P. 1999. Role of mucus trails and trail-following in the behavior and nutrition of the periwinkle Littorina littorea. Marine Ecology Progress series. 179: 247-257.
Harley CDG. 2002. Light availability indirectly limits herbivore growth and abundance in a high rocky intertidal community during the winter. Limnology and Oceanography. 47(4): 1217-1222.
Harley CDG, Anderson KM, Lebreton CAM, MacKay A, Ayala-Dias M, Chong SL, Pond LM, Maddison JHA, Hung BHC, Iversen SL. 2013. The introduction of Littorina littorea to British Columbia, Canada: potential impacts and the importance of biotic resistance by native predators. Marine Biology. 160(7): 1529-1541.
Martel A, Chia F. 1991. Drifting and dispersal of small bivalves and gastropods with direct development. Journal of Experimental Marine Biology, 150(1): 131-147.
Nwewll RC, Northcroft HR. 1967. A re-interpretation of the effect of temperature on the metabolism of certain marine invertebrates. Journal of Zoology. 151(3): 277-298.
Rickards KJ, Boulding EG. 2015. Effects of temperature and humidity on activity and microhabitat selection by Littorina subrotundata. Marine Ecology Progress Series. 537: 163-173.
Rudy P, Rudy LH, Shanks A, ButlerB. 2013. Littorina sitkana. University of Oregon Scholars' Bank. [accessed 2017 May 17]. https://scholarsbank.uoregon.edu/xmlui/bitstream/handle/1794/12904/L_sitkana.pdf?sequence=1&isAllowed=y.
Todd ME. 1964. Osmotic balance in Littorina littorea, L. littoralis, and L. saxatilis (Littorinidae). Physiological Zoology. 37(1): 33-44.
Davenport J, Davenport JL. 2005. Effects of shore height, wave exposure and geographical distance on thermal niche width of intertidal fauna. Marine Ecology Progress Series. 292: 41-50.
Davies MS, Beckwith P. 1999. Role of mucus trails and trail-following in the behavior and nutrition of the periwinkle Littorina littorea. Marine Ecology Progress series. 179: 247-257.
Harley CDG. 2002. Light availability indirectly limits herbivore growth and abundance in a high rocky intertidal community during the winter. Limnology and Oceanography. 47(4): 1217-1222.
Harley CDG, Anderson KM, Lebreton CAM, MacKay A, Ayala-Dias M, Chong SL, Pond LM, Maddison JHA, Hung BHC, Iversen SL. 2013. The introduction of Littorina littorea to British Columbia, Canada: potential impacts and the importance of biotic resistance by native predators. Marine Biology. 160(7): 1529-1541.
Martel A, Chia F. 1991. Drifting and dispersal of small bivalves and gastropods with direct development. Journal of Experimental Marine Biology, 150(1): 131-147.
Nwewll RC, Northcroft HR. 1967. A re-interpretation of the effect of temperature on the metabolism of certain marine invertebrates. Journal of Zoology. 151(3): 277-298.
Rickards KJ, Boulding EG. 2015. Effects of temperature and humidity on activity and microhabitat selection by Littorina subrotundata. Marine Ecology Progress Series. 537: 163-173.
Rudy P, Rudy LH, Shanks A, ButlerB. 2013. Littorina sitkana. University of Oregon Scholars' Bank. [accessed 2017 May 17]. https://scholarsbank.uoregon.edu/xmlui/bitstream/handle/1794/12904/L_sitkana.pdf?sequence=1&isAllowed=y.
Todd ME. 1964. Osmotic balance in Littorina littorea, L. littoralis, and L. saxatilis (Littorinidae). Physiological Zoology. 37(1): 33-44.